Most lithium-ion cathodes are manufactured using poly(vinylidene fluoride) (PVDF) as the binder and N-methyl-2-pyrrolidone (NMP) as the solvent. In practice, PVDF behaves like a passive glue: it holds particles together and survives the electrochemical environment, but it does not adapt well when particles move, crack, or strain during cycling.
While PVDF has been the industry workhorse for decades due to its high electrochemical stability and good compatibility with electrode materials, this system comes with significant tradeoffs. NMP exposure is associated with serious long-term health risks, including harm during pregnancy, and PVDF exposure has been linked to increased cancer risk and developmental harm with repeated exposure. As a result, both the EU and the US now tightly regulate NMP use in manufacturing environments.
The aqueous alternative everyone wants: CMC/SBR
Anodes already made the leap: water-based processing with CMC/SBR is the standard for graphite.
CMC (carboxymethyl cellulose) = water-soluble cellulose derivative
Thickener + dispersant + binder “skeleton”
Carboxylate groups help dispersion and interfacial bonding (hydrogen bonding + ion–dipole interactions)
SBR (styrene-butadiene rubber) = elastomeric latex in water
Mechanical compliance + flexibility + adhesion insurance
“Stops CMC from being brittle and dramatic”
Typical binder ratios in practice are often around CMC:SBR ≈ 1:2 to 1:1.5 (system-dependent, and the molecular weight + solids + drying profile matter as much as the nominal ratio).
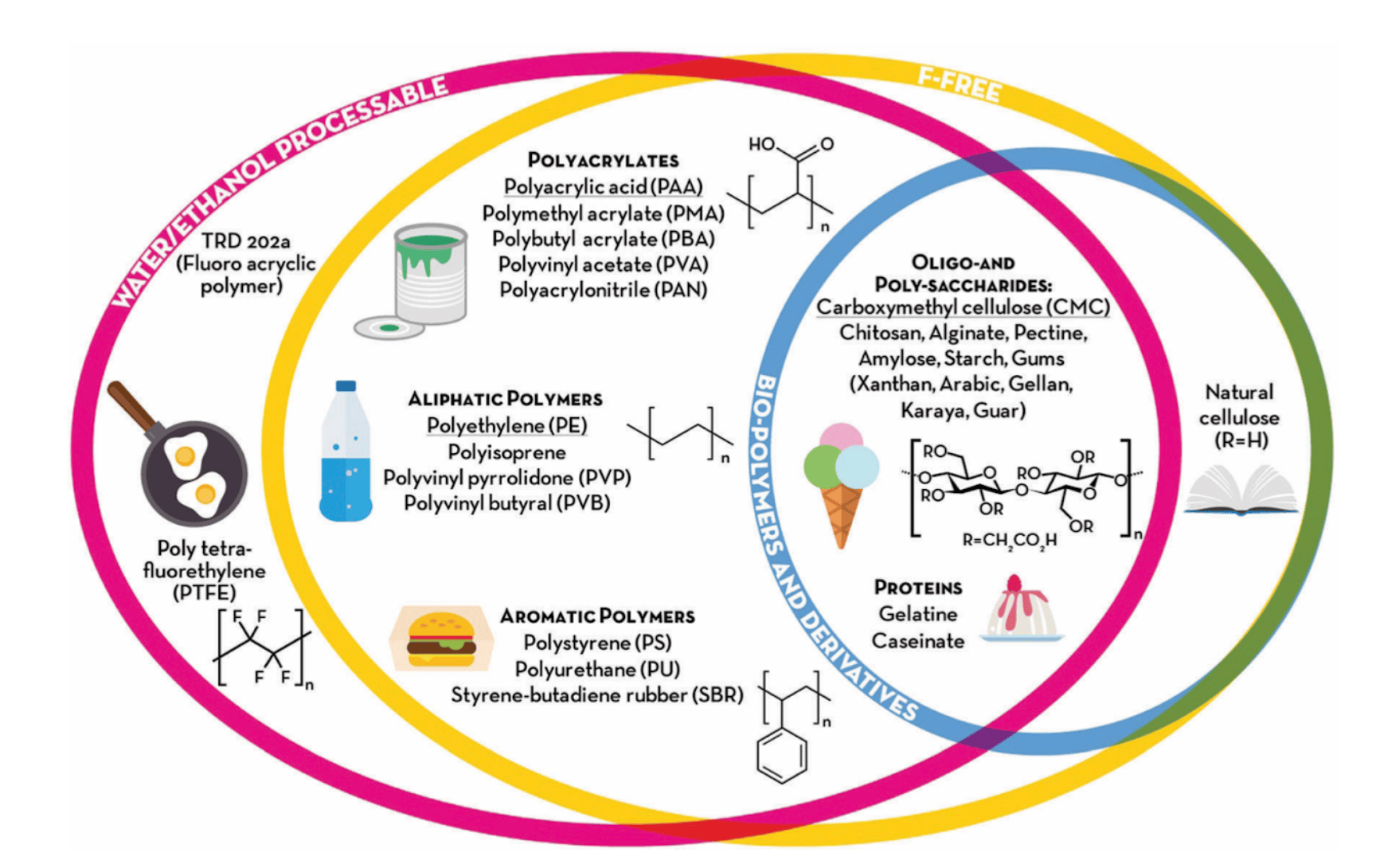
Figure 1: So you too can pretend to be a polymer scientist. Source: D. Bresser et al., Energy & Environmental Science, 2018 https://doi.org/10.1039/C8EE00640G
Why cathodes are the problem child
Trying to port CMC/SBR from anodes → cathodes is not a copy/paste.
Cathodes add new failure modes:
Water sensitivity: layered oxides can react with water → lithium leaching, surface chemistry changes, impedance growth later
pH drama: aqueous slurries can drift alkaline → Al current collector corrosion
Oxidative environment: cathodes operate at higher potentials (vs Li/Li⁺) → binder and interface need better oxidative stability than graphite
So yes, aqueous cathodes are possible. But they’re not “anode logic with a different powder.”
The 2021 roll-to-roll (R2R) paper: so close
This is what sent me: an academic roll-to-roll trial. Manufacturing in academia! Rare! Beautiful! Worth celebrating!
…but the “best” formulation was:
90% active, 1% CMC, 2% SBR, 7% carbon additive
Seven percent carbon. SEVEN.
That’s not an energy-density cathode. That’s a waste of pilot line time.
Industry people will laugh (and then quietly cry) at spending R2R time proving you can coat a formulation that’s basically designed to behave nicely.
Here’s why it matters:
Carbon content dominates slurry behavior. It changes viscosity, yield stress, percolation, % solids windows, and drying sensitivity.
If you don’t tune carbon + solids + dispersant behavior, you’re not proving “aqueous cathodes work,” you’re proving “this specific academic slurry can be made to behave on a specific coater.”
What I want to see as the real flex:
~96% active / ~3% total binder / ~1% carbon
Because that’s the direction you have to move to be taken seriously on energy density.
Also: reverse comma coater, and SBR-only didn’t adhere to Al = no cells made without CMC.
For the love of Li: publish your % solids.
If you give me “90/1/2/7” and don’t give me:
% solids
pH
mixing order + shear history
drying profile (temp, airflow, line speed)
…then you didn’t give me a process. You gave me a vibes-based formulation.
R2R does not care that it worked with a doctor blade.
Pilot lines exist to expose assumptions you didn’t know you were making.
Scaling reality: water is a thinner solvent than NMP
Water’s lower viscosity changes everything:
Harder to hold high loadings without cracking
More sensitive to drying-driven binder migration (“binder redistribution” is the quiet villain)
This is exactly why investors “only respect pouch data.” They’ve been burned. They know the lab can lie to you, politely, for months.
My hill to die on
If your binder molecular weight is missing, your formulation is a story, not a recipe.
CMC isn’t “CMC.” SBR isn’t “SBR.”
Molecular weight (and distribution), functionalization, latex particle size, and supplier-to-supplier variability can completely change:
slurry rheology
drying behavior
adhesion
crack formation
rate capability tradeoffs (yes, higher binder can help adhesion and still hurt power if it disrupts conductive networks)
And notably: multiple aqueous NMC / R2R reports don’t specify binder MW or distribution, treating binder choice like a trivial knob instead of polymer physics with consequences.
That’s why literature can “show” cracking/adhesion tradeoffs at scale while still ignoring the things that dominate pilot-line failures: web mechanics, binder lot variability, and drying-driven segregation.
TLDR: Academia do better.
Notably, none of the reported roll-to-roll or aqueous NMC studies specify the molecular weight or distribution of the CMC or SBR binders used. Binder selection is treated as a formulation variable rather than a polymer physics problem, despite the fact that molecular weight strongly influences slurry rheology, drying behavior, and adhesion at manufacturing scale. This leaves open critical questions around reproducibility, lot-to-lot variability, and transferability from lab to pilot line.
Some Resources We Love:
The aforementioned R2R paper
If you want a quick refresher on the other “battery words people fake knowing,” here’s the BBB glossary
Sources
Y. Surace et al., Batteries, 2024 https://doi.org/10.3390/batteries10030100
R. Demiryürek et al., International Journal of Energy Research, 2021 https://doi.org/10.1002/er.7171
A. C. Rolandi et al., RSC Sustainability, 2024 https://doi.org/10.1039/D4SU00098F
D. Bresser et al., Energy & Environmental Science, 2018 https://doi.org/10.1039/C8EE00640G

