Cathode Material LiNixMnyCo1−x−yO2 (NMC)
Lithium nickel manganese cobalt oxide (LiNixMnyCo1−x−yO2 or NMC) is a commonly used lithium-ion cathode material due to its improved structural stability at low SOC compared to LCO, high practical capacity, and high energy. Initial research on NMC materials featured LiNi1/3Mn1/3Co1/3O2 (NMC111) sometimes referred to as NMC333 and sometimes even NCM (don’t be that guy), where the three numbers represent the ratio of transition metals present.
NMC is a layered material, hexagonal in structure with space group R3̅m, with the structure displayed in Figure 1. NMC is understood to be a solid solution of LiCoO2, LiMnO2, LiNiO2, and LiNi½Mn½O2.
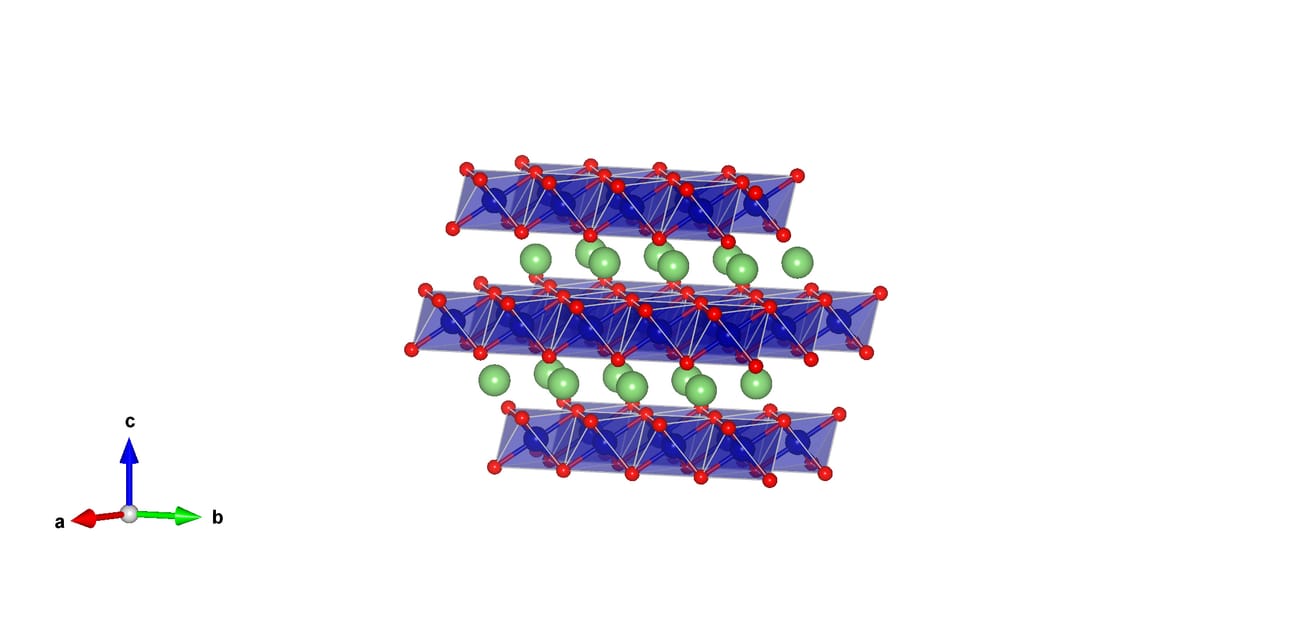
Figure 1: NMC111 structure of hexagonal R3̅m space group
Ni-rich NMC materials, where x > y, have generated significant interest in recent years due to higher capacity and further reduction of cobalt and therefore cost. LiNi0.8Mn0.1Co0.1O2 (NMC811), LiNi0.6Mn0.2Co0.2O2 (NMC622), LiNi0.5Mn0.3Co0.2O2 (NMC 532), and other variations have been predominantly used and studied.
Increasing nickel content of NMC, while positively affecting the energy density, shows increased lithium-nickel site disorder. Octahedral Ni2+ and Li+ have similar ionic radii (0.61Å and 0.76 Å respectively) allowing them to switch crystallographic sites, negatively impacting the electrochemistry by slowing lithium diffusion and reducing the rate capability of the cell. Ni-rich NMC also suffers from larger volumetric changes during delithiation, decreased thermal stability, and decreased capacity retention compared to NMC111.
Some Resources We Love :
The Quilty paper on NMC622 is one of my all time favorites for the materials science behind NMC
This Gasteiger Group publication has a figure explaining the effects of water washing in NMC811 and it’s truly 10/10

