What is a Zinc-Air Battery?
A zinc-air battery is a type of electrochemical battery composed of a zinc anode, an air cathode, and an alkaline electrolyte. These batteries are attractive because they offer extremely high specific and volumetric energy density and are relatively inexpensive. However, they also suffer from slow discharge rates and rechargeability issues due to the limitations of the air electrode. For this reason, most commercial zinc-air batteries are primary, meaning they aren’t rechargeable. Which in battery terms means: amazing once, then emotionally unavailable forever.
Applications
Today, zinc-air batteries are most used in small devices such as hearing aids, medical equipment, and pagers. Their high energy density allows them to provide long operating lifetimes in very small packages.
Chemistry
Air cathode reaction: Oxygen from the air gains electrons from the external circuit and reacts with water to form hydroxide ions in the electrolyte.
½ O2 + H2O + 2e− → 2OH−
Zinc anode reaction: Zinc metal is oxidized with hydroxide ions in the electrolyte, producing zinc oxide, water, and electrons that flow through the external circuit.
Zn + 2OH−→ ZnO + H2O + 2e−
Overall reaction: Zinc reacts with oxygen to form zinc oxide and releases electrochemical energy in the process.
Zn + ½ O2 → ZnO
Key Components
While the chemistry is relatively simple, the performance of zinc-air batteries depends heavily on how their internal components manage the movement of ions, electrons, and oxygen.
Zinc-air batteries have a zinc anode that often takes the form of gelled zinc powder and alkaline electrolyte rather than a solid zinc sheet. Using powdered zinc increases the surface area available for reaction, improving electrochemical activity and reducing internal resistance. Electrochemistry loves surface area. If batteries had dating profiles, “high surface area” would be their biggest green flag.
For the electrolyte, potassium hydroxide is the most popular choice because it’s extremely soluble and thus can be the most highly concentrated, allowing for greater ionic conductivity. The zinc and KOH are gelled together rather than simply slurried to decrease risk of leakage and eliminate the need for reservoirs to manage liquid electrolyte.
The cathode is an air electrode, which we already covered here. Because the active material is oxygen, which is external to the cell, air electrodes can be made particularly thin. This contributes to very high energy density. As a result, a thicker anode is needed in zinc-air batteries to balance the capacity of the cathode, and extremely high specific capacity values are observed, meaning they can store a large amount of charge relative to their mass. This is one of the few batteries where part of the reactant is literally free from the atmosphere, which is great for energy density and terrible for engineers who prefer controlled environments.
Additionally, a porous separator keeps the anode and cathode apart to prevent short circuiting while still allowing hydroxide ions to pass through. The separator must remain stable in the strongly alkaline electrolyte and be mechanically robust enough to resist dendrites, which are needle-like formations that can grow inside the cell. Nonwoven polymers such as polypropylene or polyethylene are commonly used because their high porosity allows efficient ion transport while preventing electrons from passing directly between the electrodes.
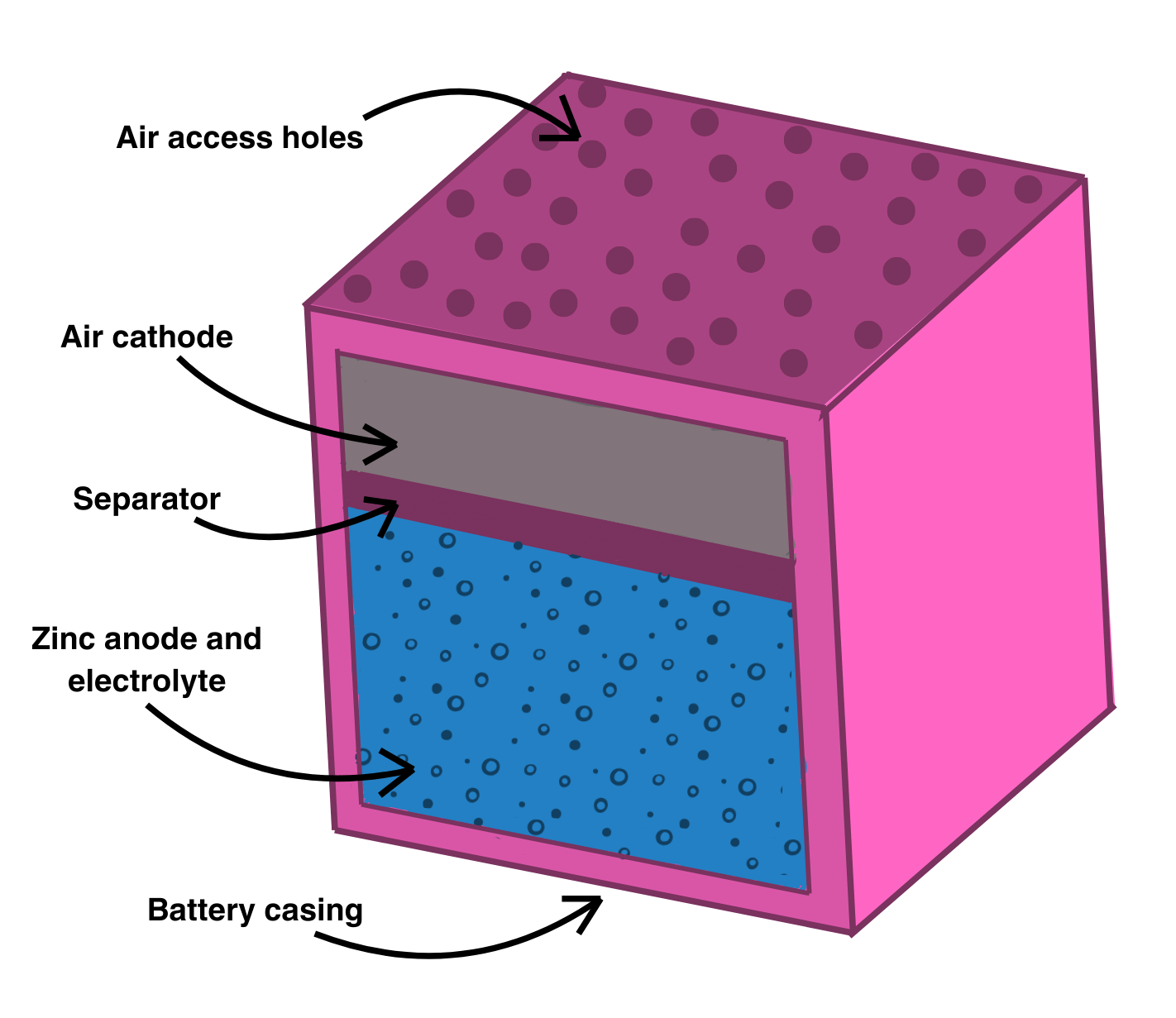
Figure 1. Schematic representation of a prismatic zinc–air battery configuration.
Pros
As mentioned earlier, zinc-air batteries have extremely high specific and volumetric energy density with values of 400 to 450 Wh/kg and 1200 to 1400 Wh/L respectively. These energy densities surpass those of primary lithium batteries and are more than 5 times greater than that of alkaline batteries. With this, zinc-air batteries are also made of inexpensive and environmentally benign materials given that zinc is affordable, air is free, and both are abundant. Another advantage is their long shelf life before activation. As long as they’re stored correctly and the air access holes are sealed before use, zinc-air batteries can be stored for extended periods without significant capacity loss. Finally, the potassium hydroxide electrolyte is water-based and inherently nonflammable, so unlike lithium-ion batteries, zinc-air cells do not carry the same risk of thermal runaway and fire.
Cons
While the air electrode is responsible for zinc-air batteries’ amazing energy density that sets it apart, the air cathode is also the culprit of most of the technology’s shortcomings. Because oxygen must diffuse into the cell through multiple layers to reach the reaction sites, and the oxygen reduction reaction is a notoriously slow one, zinc-air batteries cannot maintain large currents. This means they have very low discharge rates of anywhere from several days to a month. The air access holes that allow oxygen to reach the air electrode also make the battery sensitive to temperature and humidity and allow carbon dioxide from the air to enter the cell and degrade the electrolyte if the battery is stored uncovered. Because the atmosphere unfortunately contains more than just helpful oxygen. Additionally, as explained in depth in the Air Electrodes post, oxygen reduction (ORR) during discharge and oxygen evolution (OER) during charge are very different reactions and it’s extremely difficult to build an air electrode that can do both efficiently.
Future of Zinc-Air Batteries
For this reason, while rechargeability in zinc-air batteries isn’t impossible, most commercial zinc-air batteries are single use and not rechargeable.
Zinc-air batteries highlight one of the most important lessons in battery science: high theoretical energy density does not automatically translate into practical performance. The chemistry may look simple, but the real challenge lies in controlling oxygen transport, reaction kinetics, and electrode stability inside the cell.
And until those challenges are solved, zinc-air batteries will remain one of the most promising (and most stubborn) battery chemistries still under active research.
References
ScienceDirect. Zinc–Air Battery. Elsevier ScienceDirect Topics.
https://www.sciencedirect.com/topics/chemistry/zinc-air-battery
Zhang, J. Rechargeable Zinc–Air Batteries. In: Metal–Air Batteries: Fundamentals and Applications. Wiley, 2020. https://onlinelibrary.wiley.com/doi/10.1002/9783527837939.ch1
Li, Y., Dai, H. Recent Advances in Zinc–Air Batteries. ResearchGate figure reproduction.https://www.researchgate.net/figure/Schematic-representation-of-prismatic-zinc-air-battery-configuration_fig6_311098275

