In March 2021, a Nature Materials paper came out that altered my brain chemistry. This post is dedicated to that paper and explains the controversy behind layered metal oxide phase changes with a focus specifically on NMC. Let’s get into it.
If you missed the last newsletters on cathode materials or NMC you might want to read those first.
A very brief background and how we talk about structures
NMC (LiNiₓMnᵧCo₁₋ₓ₋ᵧO₂), LCO (LiCoO₂), and LNO (LiNiO₂) all share the same crystal structure, which belongs to the hexagonal R3̅m space group.
This structure is classified as O3 in Delmas’ notation, indicating a face-centered cubic (fcc) arrangement.
The letter "O" refers to the octahedral coordination of lithium (Li) within the structure.
The number "3" signifies the three transition metal oxide layers present in each unit cell.
The layers are arranged within the ab plane, while the c-direction is perpendicular to these layers, and the unit cell parameters satisfy a = b ≠ c.
Phase Transformations and Notation
Most phase transformations in these materials occur without disrupting the O3 stacking sequence, meaning that the O3 structure remains intact. Because of this, a secondary notation system is used to describe different phases:
Phases are labeled using a letter and a number:
The letter represents the symmetry of the unit cell.
The number indicates the order in which that phase appears during charge.
Structures that belong to the rhombohedral lattice system are typically labeled as "H" for hexagonal.
Other notable structures include:
Distorted O3 structures, which are classified as monoclinic (M).
O1 structure, belonging to space group P3m1.
Hybrid H1-3 phases, which feature alternating O1 and O3 blocks.
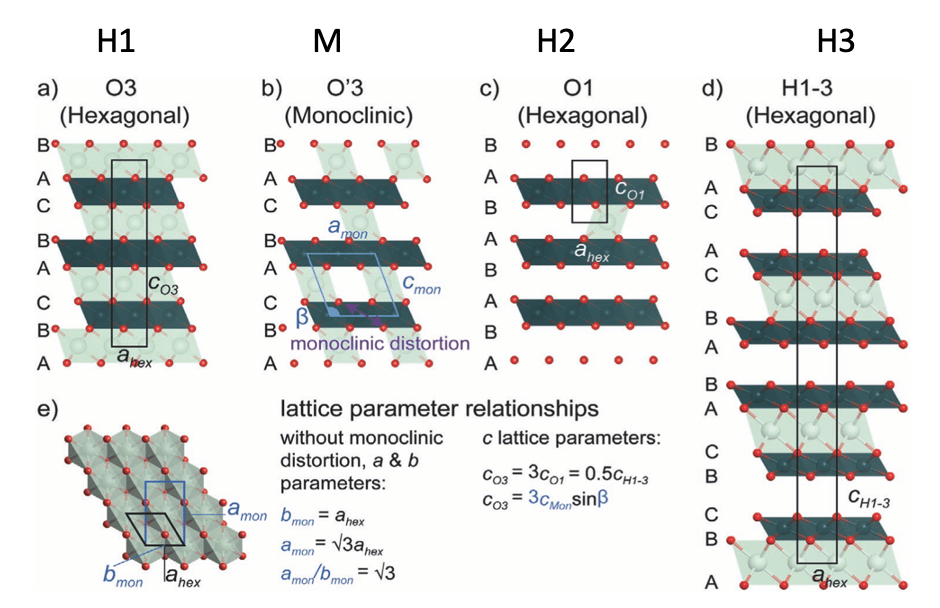
Figure 1: Illustrations of the crystal structures relevant to the layered cathodes. Unit cells are shown in black (hexagonal) or blue (monoclinic) and Li, TM, and O atoms in light green, dark green, and red respectively. [1]
LCO
In 1998, LCO was identified (through simulations) to form three different phases during cycling:
the rhombohedral form of LCO (O3)
the hexagonal form of CoO2 (O1)
a hybrid of the previous two structures (H1-3) [2, 3]
This was confirmed experimentally in 2002 with XRD data.[4]
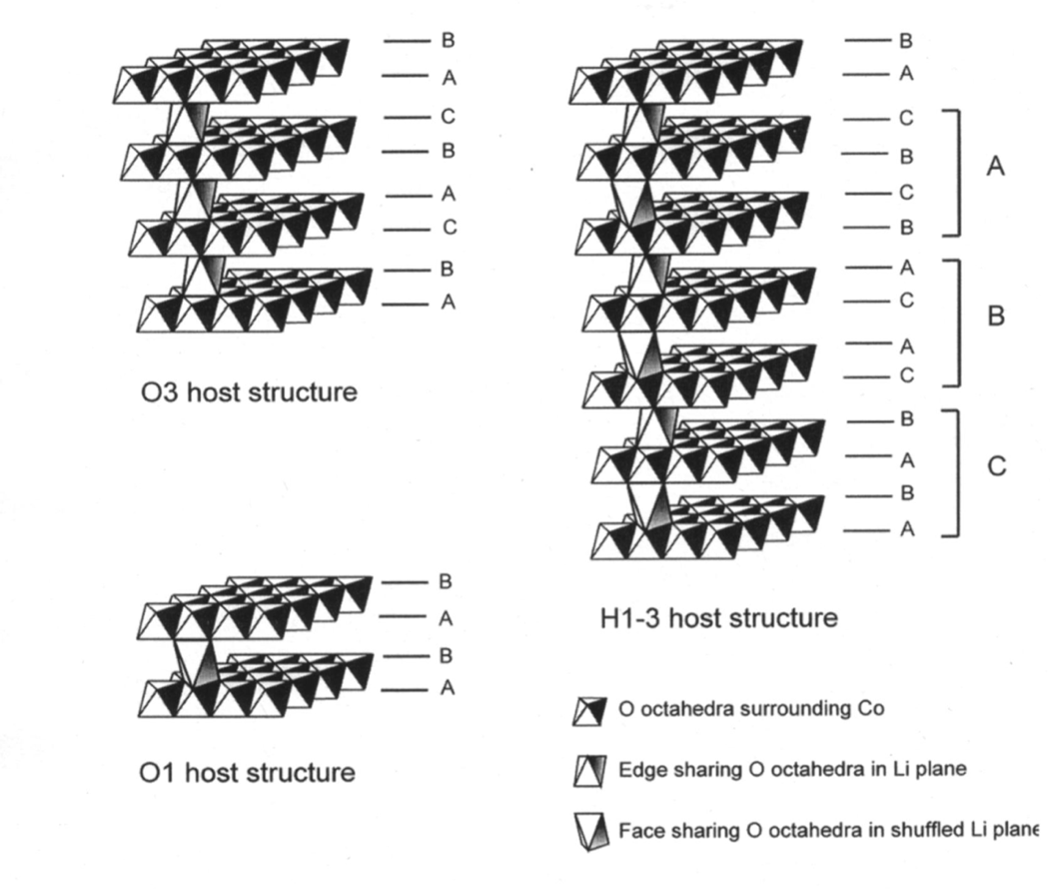
Figure 2: Schematic illustration of the three host structures O3, O1, and H1-3. Upper case letters describe the stacking of the close-packed oxygen layers.[2]
LNO
LNO was also found to have a phase change during cycling.[5-8] During charging, the c- lattice parameter increases while the a-lattice parameter decreases. This behavior is well established for LNO and isostructural NMC- and NCA-based cathodes: without the positively charged lithium ions in between the negatively charged metal oxide slabs, the like charges repel each other. Opposites attract and all that.[8]
At least four different Li1-xNiO2 phases have been described in literature: hexagonal H1, monoclinic M, hexagonal H2, and hexagonal H3 phases.[8] The sequence of phases observed for LiNiO2 is H1 → M → H2 → H3 with all phases having an O3 stacking sequence.[1]
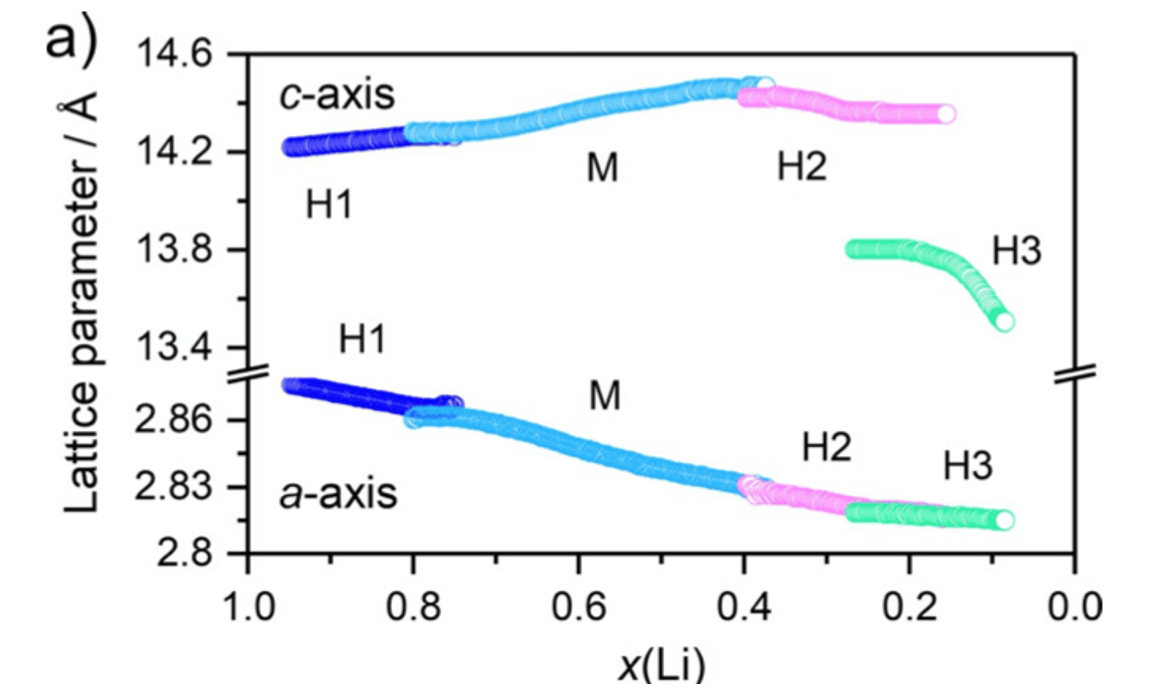
Figure 3: Evolution of a- and c-lattice parameters from Rietveld refinement analysis of operando XRD data obtained for a Li/LNO cell [8]
NMC
NMC is a solid solution of LiCoO2, LiMnO2, and LiNiO2 of hexagonal R3̅m space group.[9] Therefore, it can be reasonably assumed that phase changes occurring in LCO or LNO during cycling should also appear in NMC. RIGHT???
In 2006, a synchrotron-based in situ XRD study was done on NMC111 and NCA, and they were found to follow similar structural changes as LNO during cycling: expansion of the c-axis and contractions along the a- and b-axis during charge, and a major contraction along the c-axis with slight expansions along the a- and b-axis near the end of charge at high voltage limit. The rate cycled was C/5 for those of us keeping track.[10]
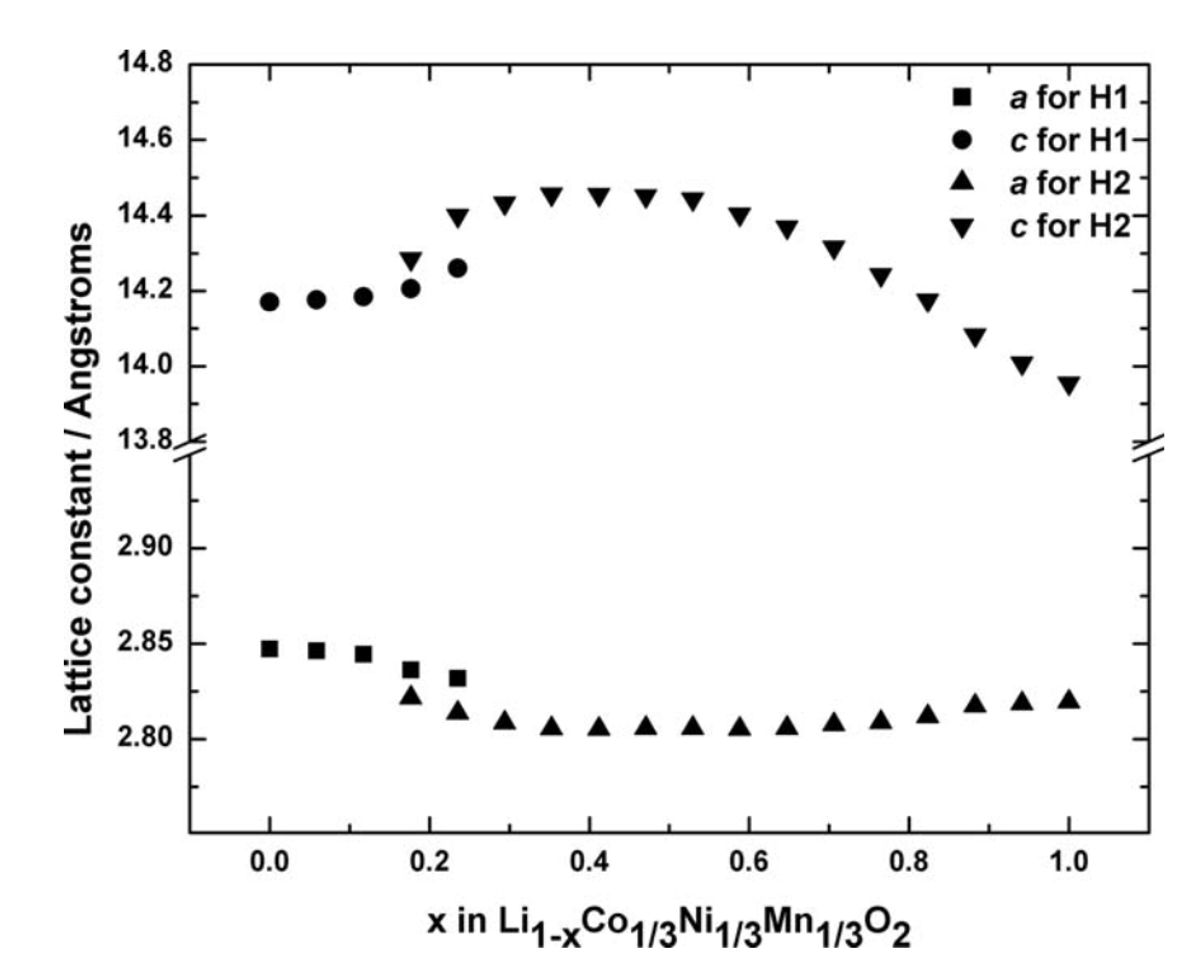
Figure 4: Variations of the lattice parameters of NMC111 as a function of x during first charge. The hexagonal phase (H1) lattice parameters (a and c) are indicated with circles and squares, and the other hexagonal phase (H2) with upper and lower triangles.[10]
Despite significant evidence of the phase transformation occurring in LNO and LCO (and some evidence in NCA and NMC), there is a lot of disagreement among NMC studies.
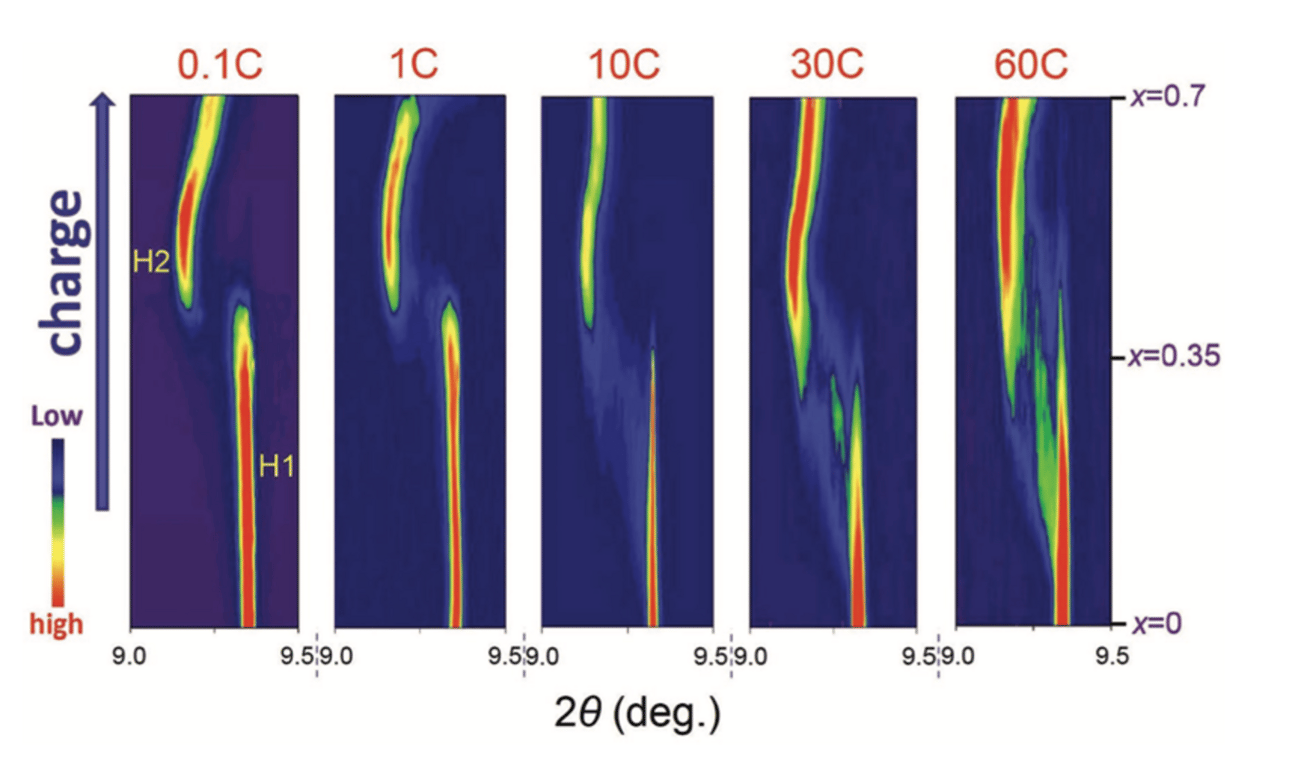
Figure 5: In situ XRD of NMC during the first charge. Contour plot of the 003 diffraction peak of NMC111 with increasing x between x = 0 and 0.7 during the first charge process at different C-rates.[11]
Zhou et al: XRD patterns of the NMC111 (LiNi1/3Mn1/3Co1/3O₂) cathode were captured during the first charge - intermediate phases emerge at high rates, but not at low rates (Figure 5).[11]
Li et al: NMC111 of small particle size was found to not have a phase change, whereas large particle size NMC did undergo a phase transition at C/100 rate.[12]
Clare Grey’s group (Xu et al) concluded that NMC811 (LiNi0.8Mn0.1Co0.1O₂), does not show a phase change as the material displays “continuous shifts with no obvious splitting.”[1]
Quilty et al. found that the two phases of NMC, H1 and H2, coexist for NMC622 (LiNi0.6Mn0.2Co0.2O₂), in all cycles – they investigated cycles 1, 2, and 100. Figure 6 shows the crystal structures of H1, H2, and H3 phases in NMC.[9] (Remember this one, it’s important).
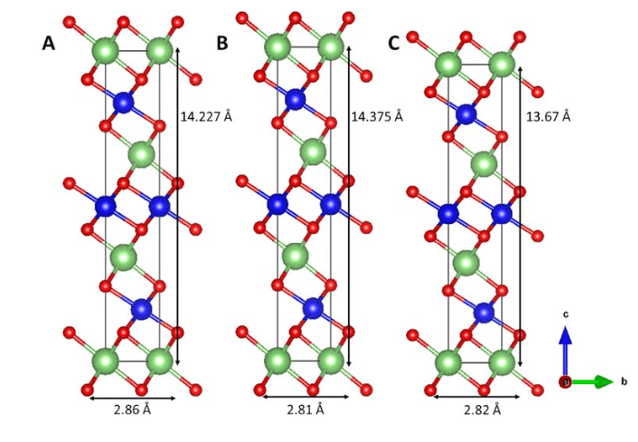
Figure 6: Crystal structures of the (A) H1, (B) H2, and (C) H3 phases of NMC. Lithium is green, oxygen is red, and the Ni/Mn/Co is blue.[9]
Controversy
In March 2021, a Nature Materials paper came out titled “Fictitious phase separation in Li layered oxides driven by electro-autocatalysis”.[13] This paper challenges traditional interpretations of phase transitions in lithium-layered oxides. I lost sleep over this paper, not going to lie to you.
The researchers observed a very clear 003 peak splitting in NMC111, a characteristic often associated with the presence of two distinct phases, H1 and H2.
However, they argue that H1 and H2 are actually the same phase, despite the apparent peak splitting in diffraction patterns.
Their findings suggest that during slow cycling, H1 can transform into H2 without any visible peak splitting, indicating a continuous structural evolution rather than a distinct phase transition.
The authors conclude that H1 and H2 must be thermodynamically identical, meaning that the previously assumed phase separation is merely an illusion ("fictitious") rather than a real physical transition.
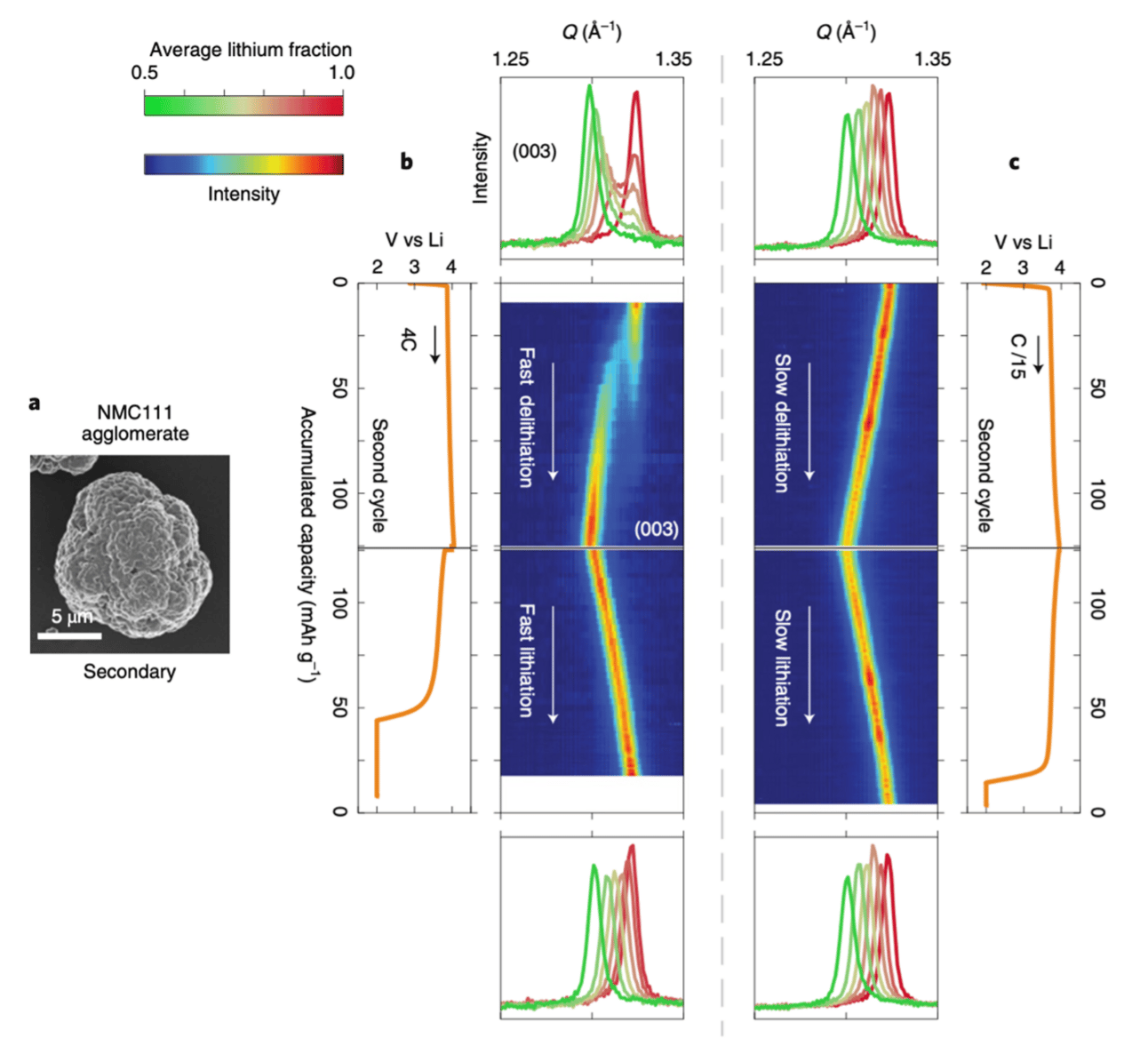
Figure 7: Phase evolution of Lix(Ni1/3Mn1/3Co1/3)O 2 during the second cycle at different cycling currents, analyzed with operando XRD. SEM images of agglomerate particles (a) and those cycled at fast (4C) (b) and slow (C/15) (c) currents. Diffraction and electrochemical data are combined to share the capacity axes. Intensity plots of the (003) peak show bifurcation during fast delithiation, while continuous shifts occur under slow conditions. Line plots of the (003) peak at selected lithium fractions reveal non-unimodal evolution during fast delithiation, contrasting with the progressive shift under slow conditions.[13]
Fictitious? Bimodal existence of NMC phases “Peak Bifurcation”
The study highlights that fictitious phase separation in lithium-layered oxides, such as NMC (LiNiₓMnᵧCo₁₋ₓ₋ᵧO₂), is a repeatable, non-equilibrium effect that results from charge redistribution rather than an actual thermodynamic phase transition. This phenomenon leads to a bimodal distribution of NMC phases, meaning that different lithium concentrations appear to coexist, even though they do not represent distinct phases.
While I would never go so far as to use the term “fictitious,” I do agree with the authors that H1, H2, and H3 are not technically unique phases: all having the same hexagonal structure with various lattice parameters. It is only the coexistence of phases and the fact that H3 tends to occur during cell failure that makes these notable. And in my continued efforts to recognize that certain verbiage is not technically correct, but continue to use it, I will keep referring to H1, H2, and H3 as “phases.” However, this nightmare-causing paper did gift us the term “peak bifurcation”, and for that, we are incredibly grateful.
Keep up the great work, Chueh group!
Sources
[1] C. Xu, P.J. Reeves, Q. Jacquet, C.P. Grey, Advanced Energy Materials, 11 (2020).
[2] A. Van der Ven, M.K. Aydinol, G. Ceder, J. Electrochem. Soc, 145 (1998) 2149-2155.
[3] A. Van der Ven, M.K. Aydinol, G. Ceder, PHYSICAL REVIEW B, 58 (1998).
[4] Z. Chen, Z. Lu, J.R. Dahn, Journal of The Electrochemical Society, 149 (2002).
[5] F.W. J.P. Peres, C. Delmas, Solid State Ionics, (1998).
[6] X.S. X.Q. Yang , J. McBreen, (1999).
[7] L. Croguennec, C. Pouillerie, A.N. Mansour, C. Delmas, Journal of Materials Chemistry, 11 (2001) 131-141.
[8] L. de Biasi, A. Schiele, M. Roca-Ayats, G. Garcia, T. Brezesinski, P. Hartmann, J. Janek, ChemSusChem, 12 (2019) 2240-2250.
[9] C.D. Quilty, D.C. Bock, S. Yan, K.J. Takeuchi, E.S. Takeuchi, A.C. Marschilok, The Journal of Physical Chemistry C, 124 (2020) 8119-8128.
[10] Z. Li, N.A. Chernova, M. Roppolo, S. Upreti, C. Petersburg, F.M. Alamgir, M.S. Whittingham, Journal of The Electrochemical Society, 158 (2011).
[11] Y.-N. Zhou, J.-L. Yue, E. Hu, H. Li, L. Gu, K.-W. Nam, S.-M. Bak, X. Yu, J. Liu, J. Bai, E. Dooryhee, Z.-W. Fu, X.-Q. Yang, Advanced Energy Materials, 6 (2016).
[12] J. Li, R. Shunmugasundaram, R. Doig, J.R. Dahn, Chemistry of Materials, 28 (2015) 162-171.
[13] J. Park, H. Zhao, S.D. Kang, K. Lim, C.C. Chen, Y.S. Yu, R.D. Braatz, D.A. Shapiro, J. Hong, M.F. Toney, M.Z. Bazant, W.C. Chueh, Nat Mater, (2021).

